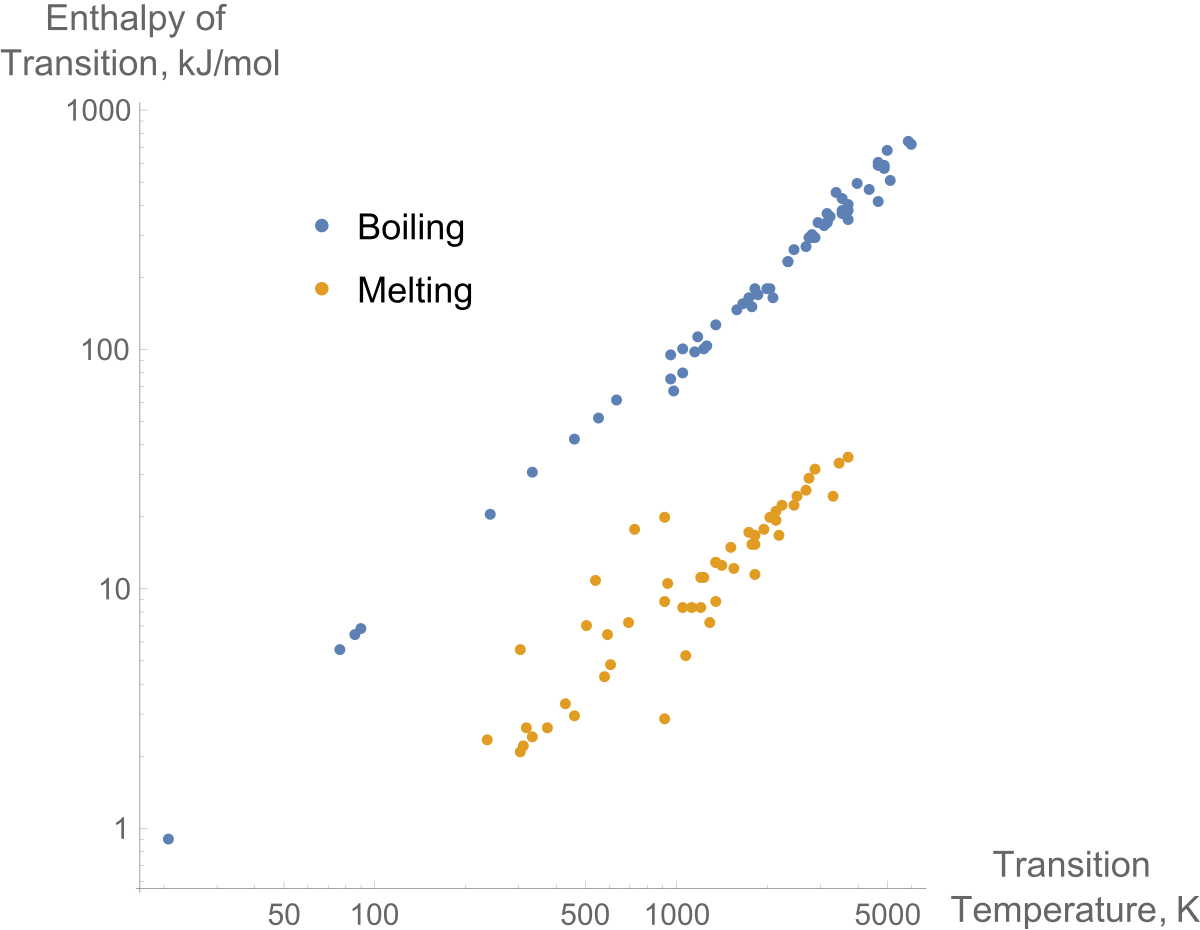
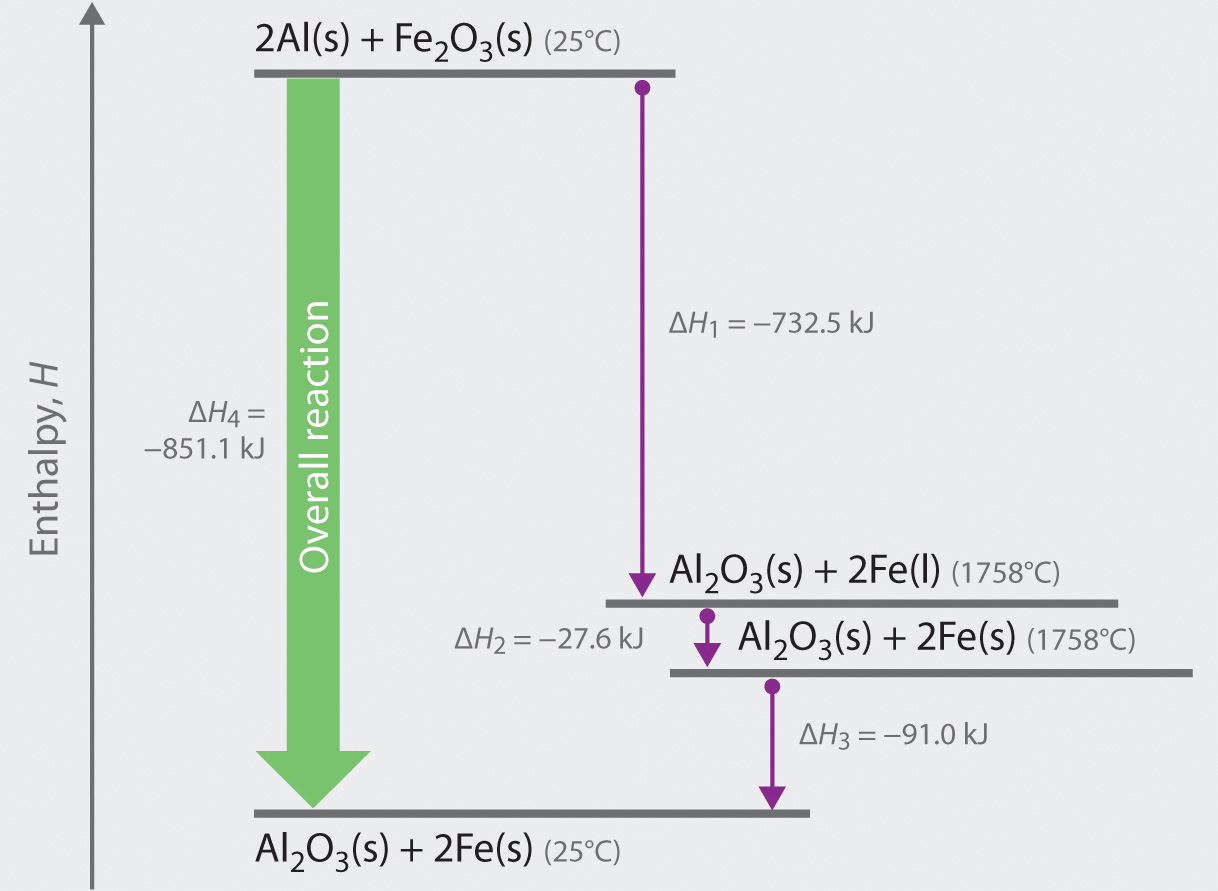
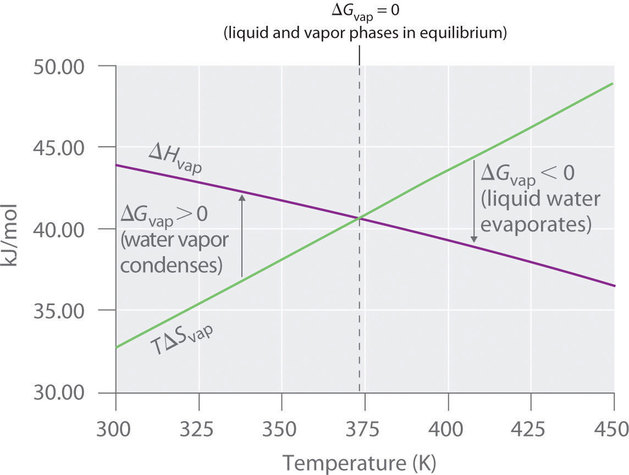
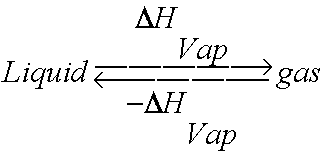Vaporization enthalpy ΔH v as a function of the temperature for CO 2.... | Download Scientific Diagram

Describe (qualitatively) how standard enthalpy and entropy of vaporization of water will change with temperature. | Homework.Study.com
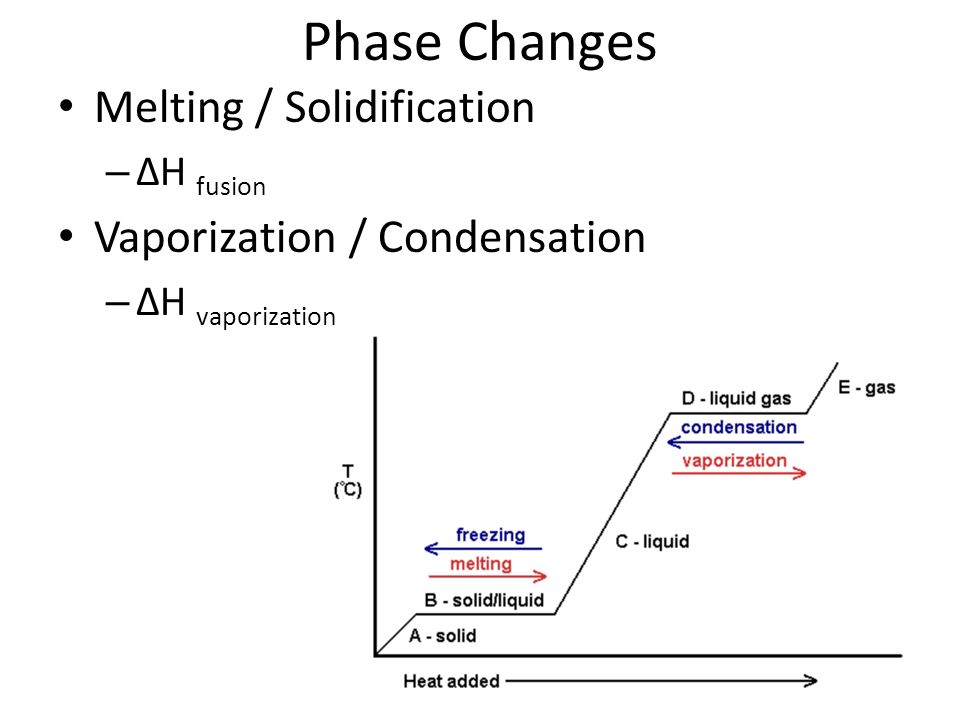
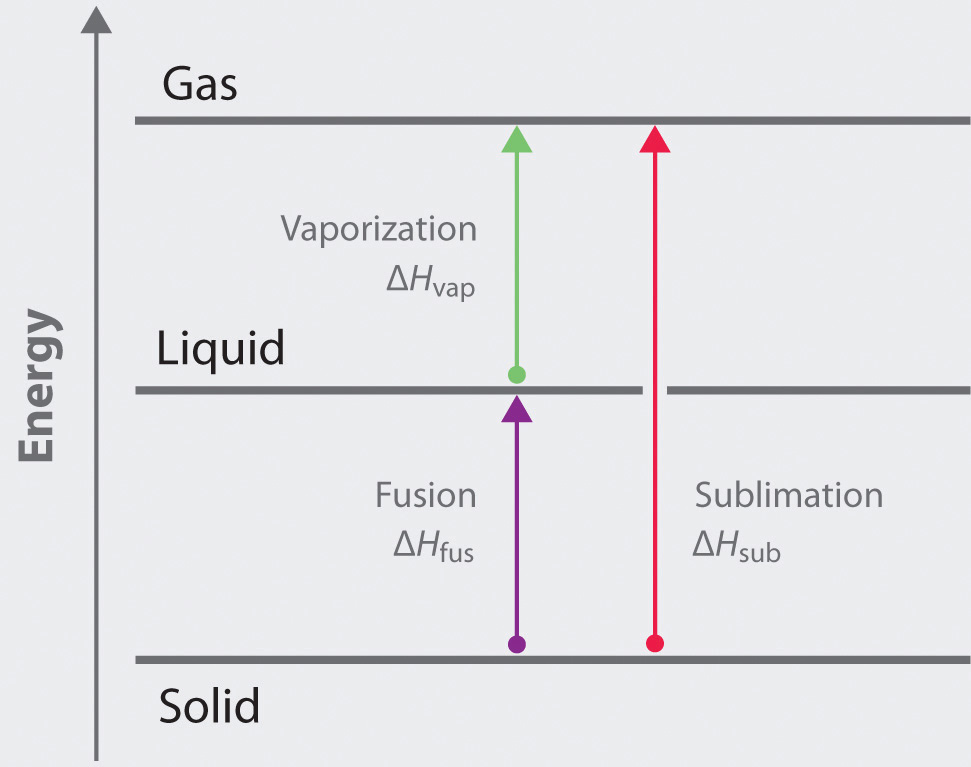
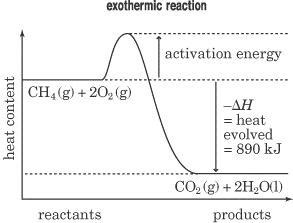
11.3 Heat in Changes of State. Warm up Is it exo- or endo- thermic???? - negative ΔH -positive ΔH -Heat as a reactant -Heat as a product -Combustion of. - ppt download

Enthalpy of vaporization as a function of temperature–values obtained... | Download Scientific Diagram

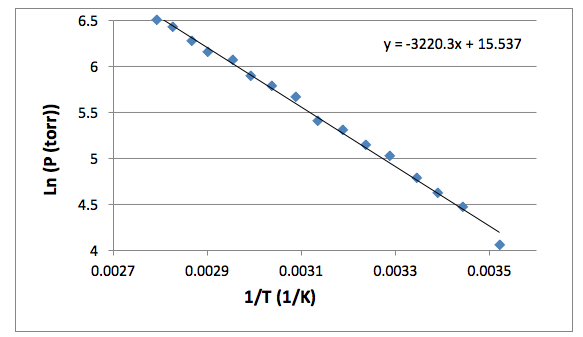
Calculate the boiling point of the liquid if its entropy of vaporization is 110JK^-1mol^-1 and the enthalpy of vaporization is 40.85kJ mol^-1 .

Enthalpy of vaporization vs. temperature for the truncated and shifted... | Download Scientific Diagram