Vapor Pressures and Thermophysical Properties of Dimethoxymethane, 1,2-Dimethoxyethane, 2-Methoxyethanol, and 2-Ethoxyethanol: Data Reconciliation and Perturbed-Chain Statistical Associating Fluid Theory Modeling | Journal of Chemical & Engineering Data

Water is the working fluid in an ideal Rankine cycle. Superheated vapor enters the turbine at 8 MPa, 480 C. The condenser pressure is 8 kPa. The net power output of the

An ideal solution at 298 K is made up of the volatile liquids A and B, for which the vapor pressures at that temperature (when pure) are PA = 125 torr and

SOLVED: The vapor pressure of benzene is found to obey the empirical equation 3229.86 K 118345 K2 16.725 T2 In(P /torr) from 298.2 K to its normal boiling point 353.24 K. Given

10.6 An ideal vapor-compression refrigeration cycle operates at steady state with Refrigerant 134a as - Brainly.com

Regions of metastable existence of ideal oxonitride solutions in an... | Download Scientific Diagram
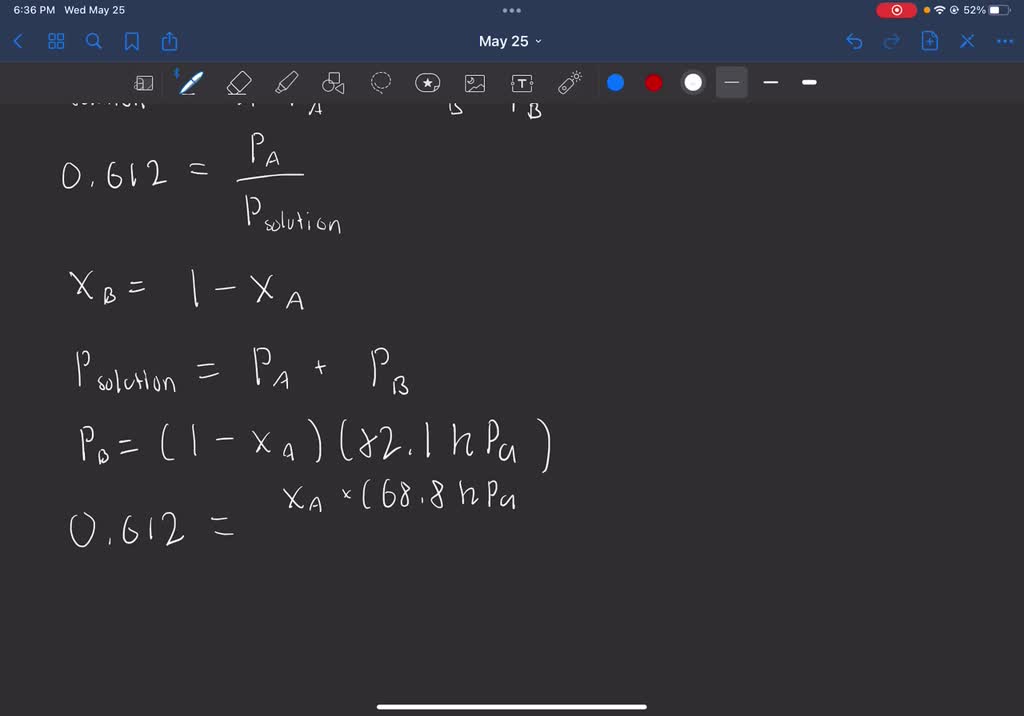
SOLVED: The vapor pressure of pure liquid A at 293 K is 68.8 kPa and that of pure liquid B is 82.[ kPa. These two compounds form ideal liquid and gaseous mixtures.

SOLVED: At 300 K (ne vapour pressure ol pure iquids and B Is 200 and 450 mmHg respactively Ine Iotal pressure ol Ihe mixture and B 350 mmHg ad the assumpllon Inat

Liquids A and B form ideal solution over the entire range of composition. At temperature, T , an equimolar binary solution of liquids A and B has vapour pressure 45 Torr. At