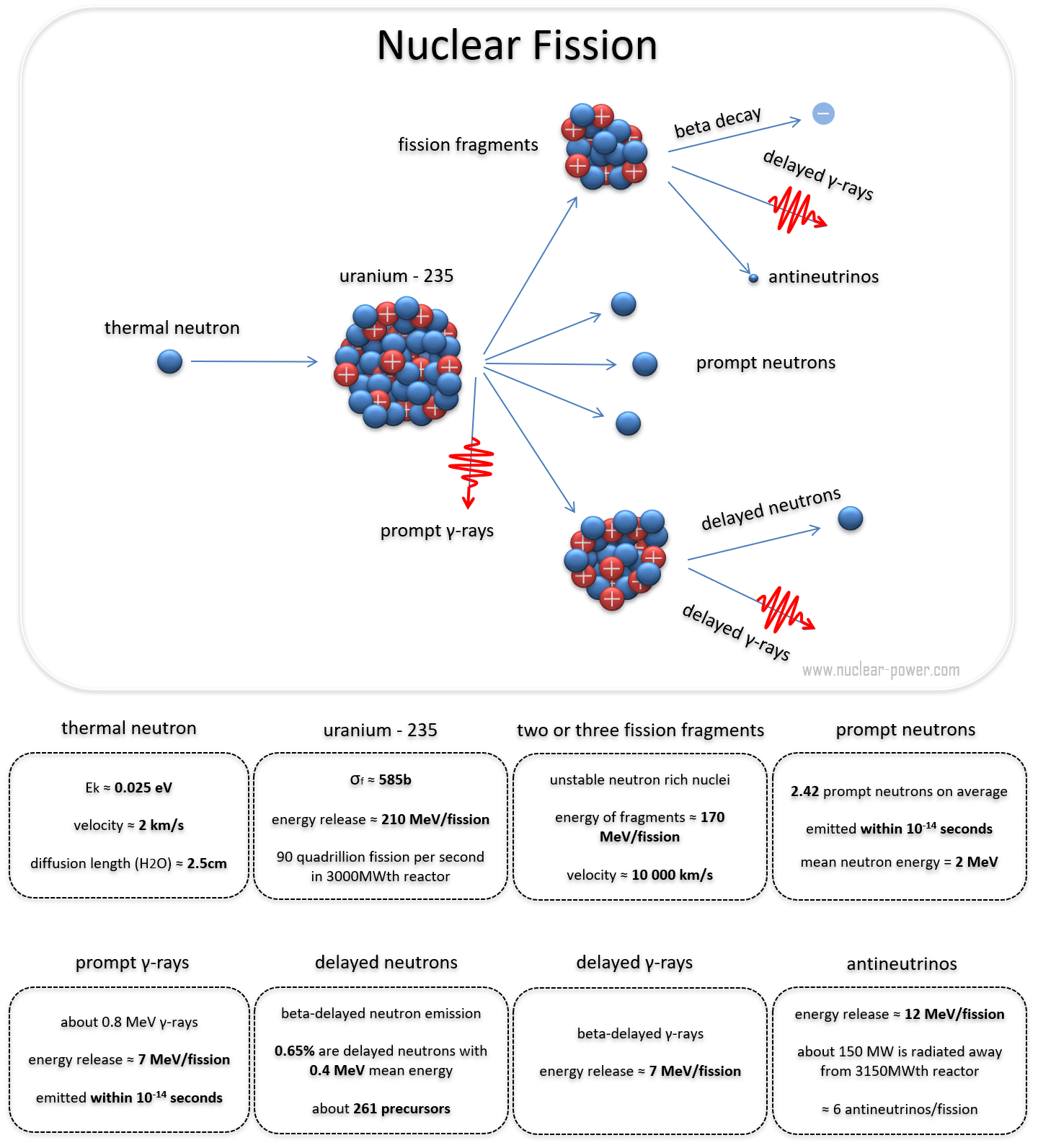
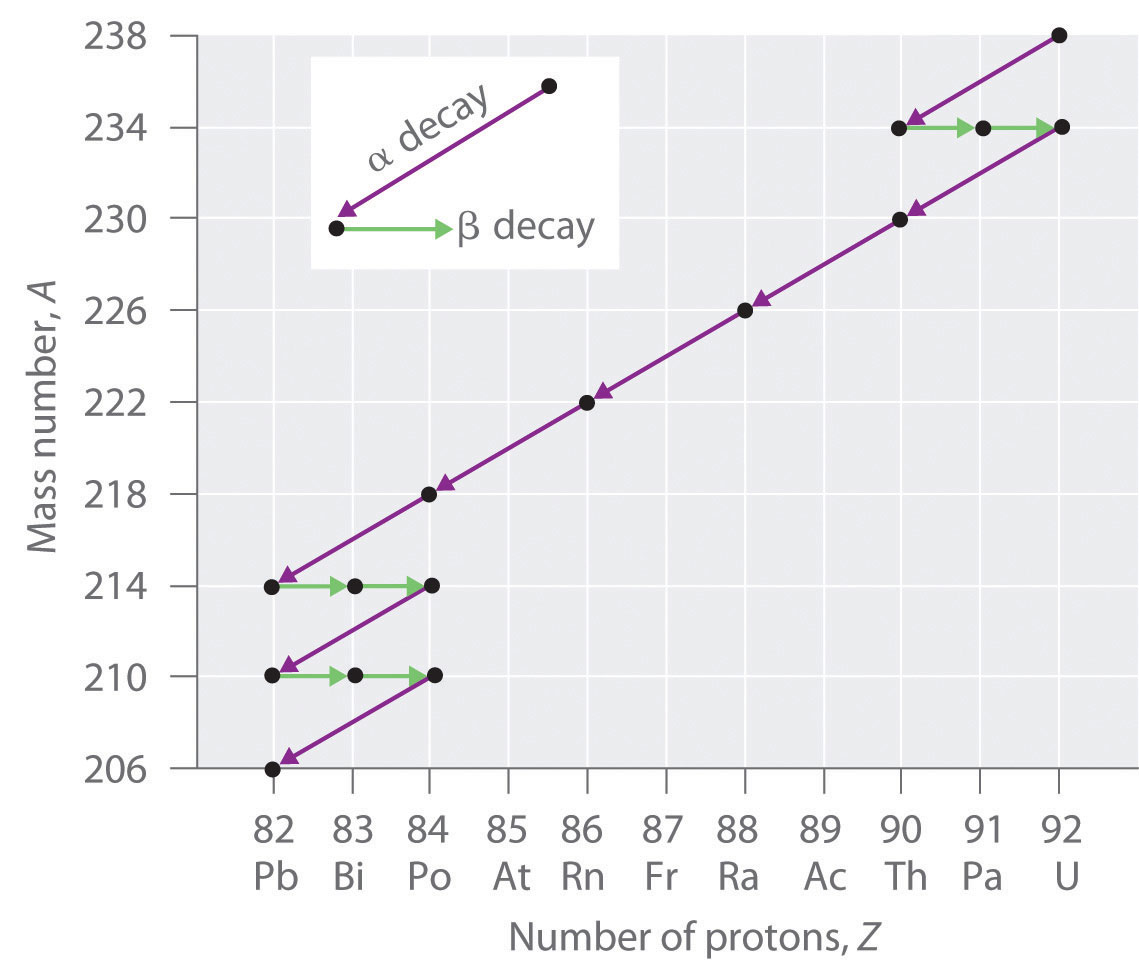
Calculate and compare the energy released by (a) Fusion of 1.0 kg of hydrogen deep within Sun (b) The fission of 1.0 kg of ^235U in a fission reactor
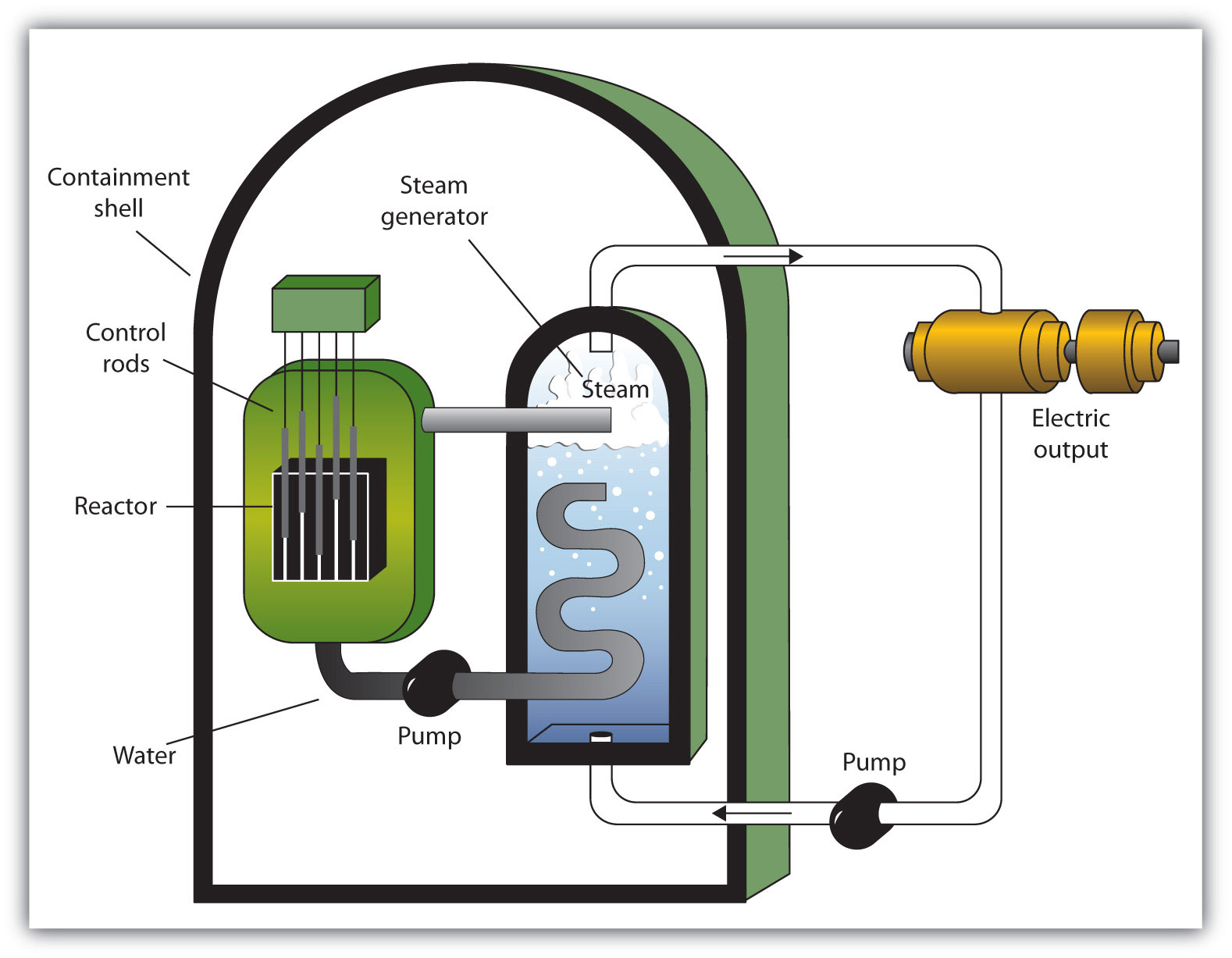
Chapter 7 - Nuclear Chemistry - CHE 105/110 - Introduction to Chemistry - Textbook - LibGuides at Hostos Community College Library
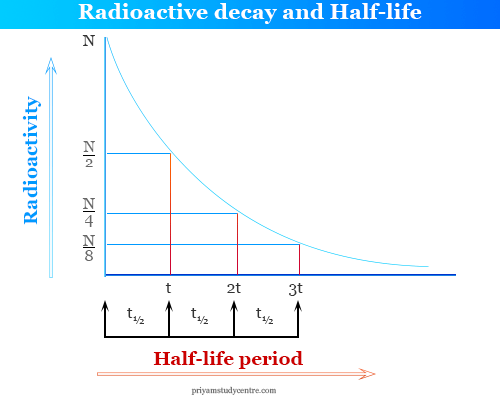
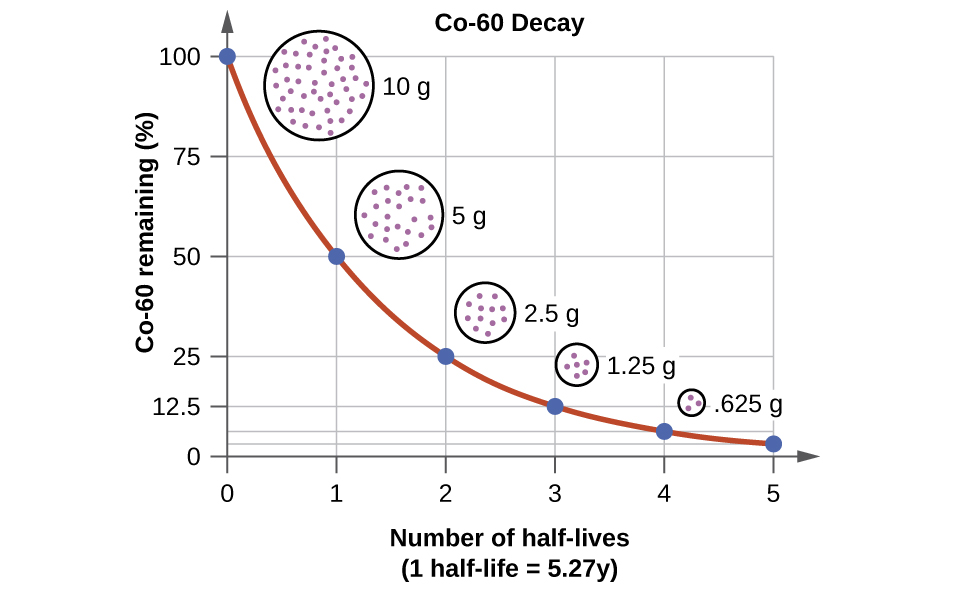
Half life of Uranium is 4.5×10^9 years. In how much time will only 10 grams be left out of 40 grams? - Quora

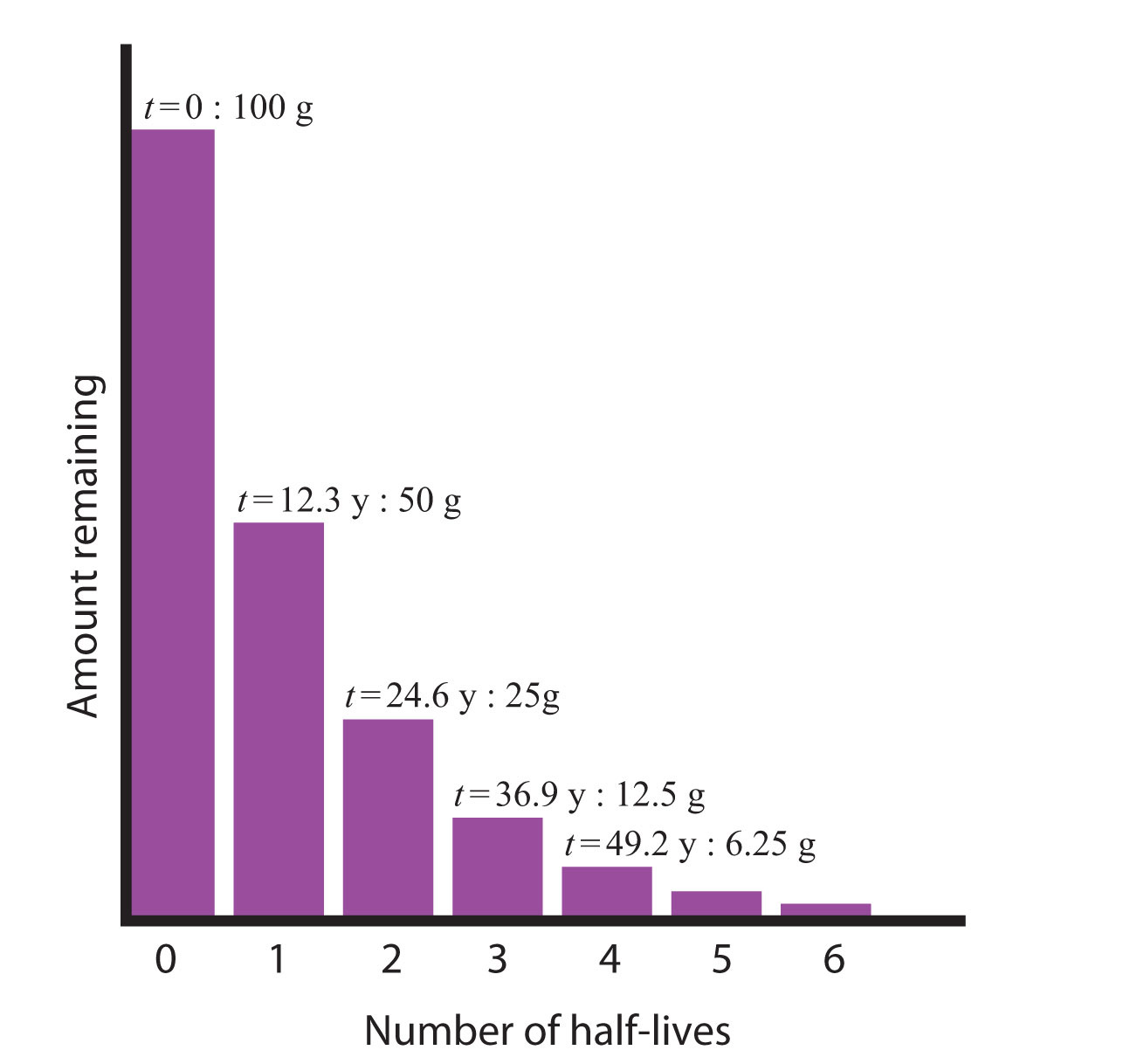
SOLVED: Uranium 235 is used as fuel for some nuclear reactors. It has a half-life of 710 million years How long- will it take 10 grams of uranium 235 to decay to