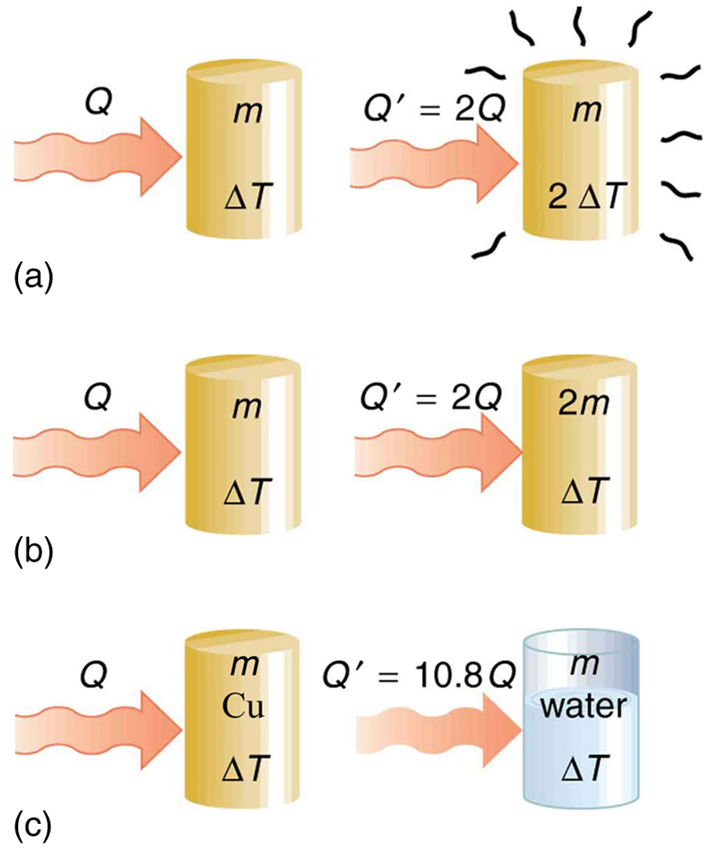
Question Video: Finding the Specific Heat Capacity of a Substance given the Change in Its Temperature and Internal Energy | Nagwa

Calculate the heat required to convert 3 kg of ice at - 12^o C kept in a calorimeter to steam at 100^o at atmospheric pressure. (Given: specific heat of ice = 2.100 ×

In a container of negligible heat capacity, 200 gm ice at 0^(@)C and 100gm steam at 100^(@)C are added to 200 gm of water that has temperature 55^(@)C. Assume no heat is